Chemical Formula of compounds and rules for writing
Table of Contents
Chemical Formula:
The symbolic representation of composition is termed as the chemical formula of a compound.
Two things are required to learn and write chemical formula:
1. Ability to write symbols, which one can attain after good practice over learning and memorising symbols.
2. Combining capacity of the element of compound or valency.
Valency :
The combining power or capacity of an element is termed as its valency.
It helps in finding out, how the atoms of an element will combine with atom(s) of the another element to form a reaction or any chemical compound.
It can be also defined as the numbers of electrons that an atom may lose or gain during sharing of electrons or any chemical reaction to achieve noble gas configuration.
For example – the sodium ion is represented as (Na+), which means that its valency is 1. Similarly, the magnesium ion is represented as (Mg2+) which means its valency is 2.
Rules For Writing a Chemical Formula:
First step is to balance the valencies or charges on the ions.
One must write the name of a metal first, if the compound consists of a metal and a non-metal. For example- sodium chloride (NaCl), copper oxide (CuO), calcium oxide (CaO) etc.
The ion is enclosed in a bracket before writing the number of ion associated to it, in case of polyatomic ion. For example – (NH 4 ) 2 SO 4
If there is single ion then no need to mention the ions in brackets. For example – NaOH.
Rules For Writing Formula Of Simple Compound :
The simplest compounds, which are made up of two different elements are called binary compounds.
Steps to write formula of a compound –
Write the symbol of the constituent elements of the compound and their respective valencies or charges as shown below.
Crossover the valencies of the elements.
Write down the symbols with the valencies or charges which you get after crossing over.
For example –
1. The formula of water :
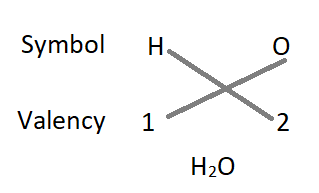 – The formula of the compound will be H2O.
– The formula of the compound will be H2O.
2. The formula of carbon tetrachloride :
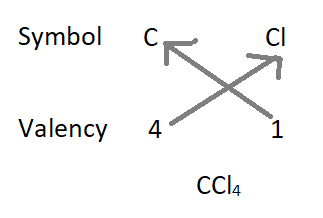 – Formula of the compound will be CCl4
– Formula of the compound will be CCl4
3. Formula of aluminium oxide :
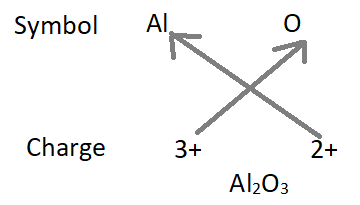 – Formula of the compound will be Al2O3
– Formula of the compound will be Al2O3
4. Formula of ammonium sulfate:
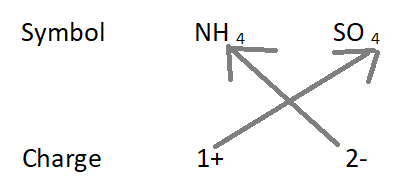 The formula of the compound will be (NH 4 ) 2 SO 4
The formula of the compound will be (NH 4 ) 2 SO 4
Categorized Under:
More Study Notes on this Topic
Ready for More?
Test your knowledge with these related materials.
📺 Watch & Learn
Visual explanations and related video classes for better understanding.
🎥 Video Resources
Naming Ionic and Molecular Compounds | How to Pass Chemistry

Naming Ionic and Molecular Compounds | How to Pass Chemistry

Writing Chemical Formulas For Ionic Compounds

Writing Chemical Formulas For Covalent Molecular Compounds

1.29-Writing Chemical Formulae Of The Compounds, class 11 some basic concept of chemistry

Naming Covalent Molecular Compounds

How to Draw Skeletal Structure or Bond-Line Notation for Organic Molecules