Energy demand and supply
The organisms possess variable energy demands, hence the supply (input) is also equally variable. The consumed metabolic fuel may be burnt (oxidized to CO2 and H2O) or stored to meet the energy requirements as per the body needs. ATP serves as the energy currency of the cell in this process. The humans possess enormous capacity for food consumption. It is estimated that one can consume as much as 100 times his/her basal requirements! Obesity, a disorder of over- nutrition mostly prevalent in affluent societies, is primarily a consequence of overconsumption.Integration of major metabolic pathways of energy metabolism
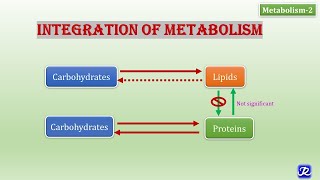
1. Glycolysis : The degradation of glucose to pyruvate (lactate under anaerobic condition) generates 8 ATP. Pyruvate is converted to acetyl CoA.2. Fatty acid oxidation : Fatty acids undergo sequential degradation with a release of 2-carbon fragment, namely acetyl CoA. The energy is trapped in the form of NADH and FADH2.
3. Degradation of amino acids : Amino acids, particularly when consumed in excess than required for protein synthesis, are degraded and utilized to meet the fuel demands of the body. The glucogenic amino acids can serve as precursors for the synthesis of glucose via the formation of pyruvate or intermediates of citric acid cycle. The ketogenic amino acids are the precursors for acetyl CoA.
4. Citric acid cycle : Acetyl CoA is the key and common metabolite, produced from different fuel sources (carbohydrates, lipids, amino acids). Acetyl CoA enters citric acid (Krebs) cycle and gets oxidized to CO2. Thus, citric acid cycle is the final common metabolic pathway for the oxidation of all foodstuffs. Most of the energy is trapped in the form of NADH and FADH2.
5. Oxidative phosphorylation : The NADH and FADH2, produced in different metabolic pathways, are finally oxidized in the electron transport chain (ETC). The ETC is coupled with oxidative phosphorylation to generate ATP.
6. Hexose monophosphate shunt : This pathway is primarily concerned with the liberation of NADPH and ribose sugar. NADPH is utilized for the biosynthesis of several compounds, including fatty acids. Ribose is an essential component of nucleotides and nucleic acids (Note : DNA contains deoxyribose).
7. Gluconeogenesis : The synthesis of glucose from non-carbohydrate sources constitutes gluconeogenesis. Several compounds (e.g. pyruvate, glycerol, amino acids) can serve as precursors for gluconeogenesis.
8. Glycogen metabolism : Glycogen is the storage form of glucose, mostly found in liver and muscle. It is degraded (glycogenolysis) and synthesized (glycogenesis) by independent pathways. Glycogen effectively serves as a fuel reserve to meet body needs, for a brief period (between meals).
Regulation of metabolic pathways
The metabolic pathways, in general, are controlled by four different mechanisms1. The availability of substrates
2. Covalent modification of enzymes
3. Allosteric regulation
4. Regulation of enzyme synthesis
ORGAN SPECIALIZATION AND METABOLIC INTEGRATION
The various tissues and organs of the body work in a well-coordinated manner to meet its metabolic demands. The major organs along with their most important metabolic functions, in a well-fed absorptive state (usually 2-4 hours after food consumption), are described. Liver
The liver is specialized to serve as the body’s central metabolic clearing house. It processes and distributes the nutrients to different tissues for utilization. After a meal, the liver takes up the carbohydrates, lipids and most of the amino acids, processes them and routes to other tissues. The major metabolic functions of liver, in an absorptive state, are listed
1. Carbohydrate metabolism : Increased glycolysis, glycogenesis and hexose mono- phosphate shunt and decreased gluconeogenesis.
2. Lipid metabolism : Increased synthesis of fatty acids and triacylglycerols.
3. Protein metabolism : Increased degradation of amino acids and protein synthesis.
Adipose tissue
Adipose tissue is regarded as the energy storage tissue. As much as 15 kg. of triacylglycerol (equivalent to 135,000 Cal) is stored in a normal adult man. The major metabolic functions of adipose tissue in an absorptive state are listed here.
1. Carbohydrate metabolism : The uptake of glucose is increased. This follows an increase in glycolysis and hexose monophosphate shunt.
2. Lipid metabolism : The synthesis of fatty acids and triacylglycerols is increased. The degradation of triacylglycerols is inhibited.
Skeletal muscle
The metabolism of skeletal muscle is rather variable depending on its needs. For instance, the resting muscle of the body utilizes about 30% of body’s oxygen consumption. However, during strenuous exercise, this may be as high as 90%. The important metabolic functions of skeletal muscle in an absorptive state are listed.
1. Carbohydrate metabolism : The uptake of glucose is higher, and glycogen synthesis is increased.
2. Lipid metabolism : Fatty acids taken up from the circulation are also important fuel sources for the skeletal muscle.
3. Protein metabolism : Incorporation of amino acids into proteins is higher.
Brain
The human brain constitutes about 2% of the body’s weight. But it utilizes as much as 20% of the oxygen consumed by the body. Being a vital organ, special priority is given to the metabolic needs of the brain.
1. Carbohydrate metabolism : In an absorptive state, glucose is the only fuel source to the brain. About 120 g of glucose is utilized per day by an adult brain. This constitutes about 60% of the glucose consumed by the body at est. It is estimated that about 50% of the energy consumed by brain is utilized by plasma membrane Na+-K+-ATPase to maintain membrane potential required for nerve impulse transmission.
2. Lipid metabolism : The free fatty acids cannot cross the blood-brain barrier, hence their contribution for the supply of energy to the brain is insignificant. Further, in a fed state, ketone bodies are almost negligible as fuel source to the brain. However, brain predominantly depends on ketone bodies during prolonged starvation .
METABOLISM IN STARVATION
Starvation may be due to food scarcity or the desire to rapidly lose weight or certain clinical conditions (e.g. surgery, burns etc.). Starvation is a metabolic stress which imposes certain metabolic compulsions on the organism. The metabolism is reorganized to meet the new demands of starvation.The survival time of an individual on starvation is mostly dependent on his/her fat stores. And for this reason, obese individuals can survive longer than lean individuals without consuming food.
Protein is basically a structural constituent, mostly present in the muscle. However, during starvation, protein can also meet the fuel demands of the body. It is estimated that about 1/3rd of the body’s protein can be utilized towards energy needs without compromising the vital functions.
Starvation is associated with a decrease in insulin level and an increase in glucagon. The metabolic changes during starvation are discussed with reference to the major organs/ tissues.
Liver in starvation
1. Carbohydrate metabolism : An important function of liver is to act as a blood glucose buffering organ. The action of liver is to suit the metabolic needs of the body. During starvation, increased gluconeogenesis and elevated glycogen degradation furnish glucose to the needy tissues (mostly brain).2. Lipid metabolism : Fatty acid oxidation is increased with an elevated synthesis of ketone bodies. This is due to the fact that TCA (Krebs) cycle cannot cope up with the excess production of acetyl CoA, hence the latter is diverted for ketone body synthesis.
Ketone bodies (primarily -hydroxybutyrate) effectively serve as fuel source for the peripheral tissues. The brain slowly adapts itself to use ketone bodies. Thus, after a 3-day fast, about 1/3rd of the brain’s fuel demands are met by ketone bodies, while, after 40 days’ starvation, they countribute to about 70% of energy needs.
Adipose tissue in starvation
1. Carbohydrate metabolism : Glucose uptake and its metabolism are lowered.2. Lipid metabolism : The degradation of triacylglycerol is elevated, leading to an increased release of fatty acids from the adipose tissue which serve as fuel source for various tissues (brain is an exception). The glycerol liberated in lipolysis serves as a precursor for glucose synthesis by liver. The synthesis of fatty acids and triacylglycerols is totally stopped in adipose tissue.
Skeletal muscle in starvation
1. Carbohydrate metabolism : Glucose uptake and its metabolism are very much depressed.2. Lipid metabolism : Both fatty acids and ketone bodies are utilized by the muscle as fuel source. However, on prolonged starvation beyond 3 weeks, the muscle adapts to exclusively utilize fatty acids. This further increases the level of ketone bodies in the circulation.
3. Protein metabolism : During the early period of starvation, muscle proteins are degraded to liberate the amino acids which are effectively utilized by the liver for glucose synthesis (gluconeogenesis). On prolonged starvation, however, protein breakdown is reduced.
Brain in starvation
As already stated, glucose is the preferred fuel source by brain. During the first 2 weeks of starvation, the brain is mostly dependent on glucose, supplied by liver gluconeogenesis. This, in turn, is dependent on the amino acids released from the muscle protein degradation. Starvation beyond 3 weeks generally results in a marked increase in plasma ketone bodies. By this time, the brain adapts itself to depend on ketone bodies for the energy needs.The biochemical changes that occur during starvation are such that an adequate supply of fuel molecules is maintained to various tissues to meet the energy demands. This is a natural adaptation for the survival of the organism.
.png)

















.jpg)
.png)







 संज्ञा
संज्ञा