A sort of gene therapy protects nervus opticus cells and preserves vision in mouse models of glaucoma, consistent with research supported by National Institute of Health (NIH’s) National Eye Institute.
The findings suggest how forward for developing neuroprotective therapies for glaucoma, a number one explanation for visual defect and blindness.
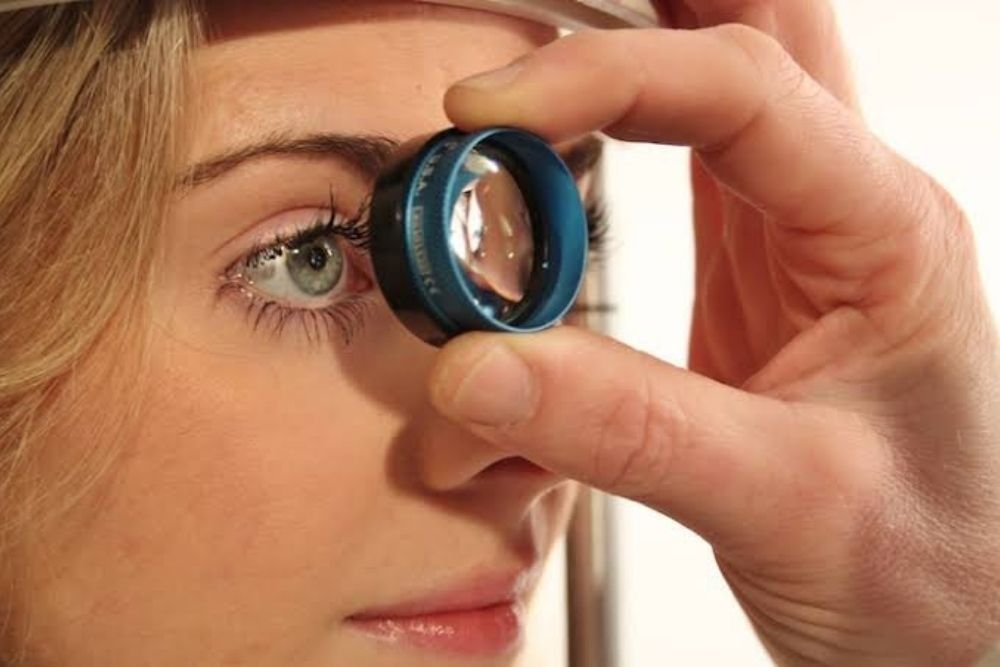
Glaucoma results from irreversible neurodegeneration of the nervus opticus , the bundle of axons from retinal ganglion cells that transmits signals from the attention to the brain to supply vision.
Available therapies slow vision loss by lowering elevated eye pressure; however some glaucoma progresses to blindness despite normal eye pressure. Neuroprotective therapies would be a breakthrough , meeting the requirements of patients who lack treatment options.
“Our study is that the first to point out that activating the CaMKII pathway helps protect retinal ganglion cells from a spread of injuries and in multiple glaucoma models,” said the study’s lead investigator, Bo Chen, Ph.D., professor of ophthalmology and neuroscience at the Icahn School of drugs at Sinai in ny City.
The CaMKII (calcium/calmodulin-dependent protein kinase II) pathway regulates key cellular processes and functions throughout the body, including retinal ganglion cells within the eye. Yet the precise role of CaMKII in retinal ganglion cell health isn't well understood. Inhibition of CaMKII activity, for instance , has been shown to be either protective or detrimental to retinal ganglion cells, counting on the conditions.
Using an antibody marker of CaMKII activity, Chen’s team discovered that CaMKII pathway signaling was compromised whenever retinal ganglion cells were exposed to toxins or trauma from a crush injury to the nervus opticus , suggesting a correlation between CaMKII activity and retinal ganglion cell survival. Searching for ways to intervene, they found that activating the CaMKII pathway with gene therapy proved protective to the retinal ganglion cells.
Administering the gene therapy to mice just before the toxic insult (which initiates rapid damage to the cells), and just after nervus opticus crush (which causes slower damage), increased CaMKII activity and robustly protected retinal ganglion cells.
Among gene therapy-treated mice, 77% of retinal ganglion cells survived 12 months after the toxic insult compared with 8% on top of things mice.
Six months following nervus opticus crush, 77% of retinal ganglion cells had survived versus 7% in controls.
Similarly, boosting CaMKII activity via gene therapy proved protective of retinal ganglion cells in glaucoma models supported elevated eye pressure or genetic deficiencies.
Increasing retinal ganglion cell survival rates translated into greater likelihood of preserved visual function, consistent with cell activity measured by electroretinogram and patterns of activity within the visual area .
Three vision-based behavioral tests also confirmed sustained visual function among the treated mice. during a visual water task, the mice were trained to swim toward a submerged platform on the idea of visual stimuli on a computer monitor.
Depth perception was confirmed by a visible cliff test supported the mouse’s innate tendency to step to the shallow side of a cliff. Lastly, a looming test determined that treated mice were more apt to reply defensively (by hiding, freezing or tail rattling) when shown an overhead stimulus designed to simulate a threat, compared with untreated mice.
“If we make retinal ganglion cells more resistant and tolerant to the insults that cause necrobiosis in glaucoma, they could be ready to survive longer and maintain their function,” Chen concluded.
The findings suggest how forward for developing neuroprotective therapies for glaucoma, a number one explanation for visual defect and blindness.
Glaucoma results from irreversible neurodegeneration of the nervus opticus , the bundle of axons from retinal ganglion cells that transmits signals from the attention to the brain to supply vision.
Available therapies slow vision loss by lowering elevated eye pressure; however some glaucoma progresses to blindness despite normal eye pressure. Neuroprotective therapies would be a breakthrough , meeting the requirements of patients who lack treatment options.
“Our study is that the first to point out that activating the CaMKII pathway helps protect retinal ganglion cells from a spread of injuries and in multiple glaucoma models,” said the study’s lead investigator, Bo Chen, Ph.D., professor of ophthalmology and neuroscience at the Icahn School of drugs at Sinai in ny City.
The CaMKII (calcium/calmodulin-dependent protein kinase II) pathway regulates key cellular processes and functions throughout the body, including retinal ganglion cells within the eye. Yet the precise role of CaMKII in retinal ganglion cell health isn't well understood. Inhibition of CaMKII activity, for instance , has been shown to be either protective or detrimental to retinal ganglion cells, counting on the conditions.
Using an antibody marker of CaMKII activity, Chen’s team discovered that CaMKII pathway signaling was compromised whenever retinal ganglion cells were exposed to toxins or trauma from a crush injury to the nervus opticus , suggesting a correlation between CaMKII activity and retinal ganglion cell survival. Searching for ways to intervene, they found that activating the CaMKII pathway with gene therapy proved protective to the retinal ganglion cells.
Administering the gene therapy to mice just before the toxic insult (which initiates rapid damage to the cells), and just after nervus opticus crush (which causes slower damage), increased CaMKII activity and robustly protected retinal ganglion cells.
Among gene therapy-treated mice, 77% of retinal ganglion cells survived 12 months after the toxic insult compared with 8% on top of things mice.
Six months following nervus opticus crush, 77% of retinal ganglion cells had survived versus 7% in controls.
Similarly, boosting CaMKII activity via gene therapy proved protective of retinal ganglion cells in glaucoma models supported elevated eye pressure or genetic deficiencies.
Increasing retinal ganglion cell survival rates translated into greater likelihood of preserved visual function, consistent with cell activity measured by electroretinogram and patterns of activity within the visual area .
Three vision-based behavioral tests also confirmed sustained visual function among the treated mice. during a visual water task, the mice were trained to swim toward a submerged platform on the idea of visual stimuli on a computer monitor.
Depth perception was confirmed by a visible cliff test supported the mouse’s innate tendency to step to the shallow side of a cliff. Lastly, a looming test determined that treated mice were more apt to reply defensively (by hiding, freezing or tail rattling) when shown an overhead stimulus designed to simulate a threat, compared with untreated mice.
“If we make retinal ganglion cells more resistant and tolerant to the insults that cause necrobiosis in glaucoma, they could be ready to survive longer and maintain their function,” Chen concluded.
.png)










.jpg)
.png)







 Registration Open For IGNOU's 33rd Convocation Programme 2020
Registration Open For IGNOU's 33rd Convocation Programme 2020  DSSSB : List of exams scheduled on 01, 09, 10, 16 and 17 of October 2021
DSSSB : List of exams scheduled on 01, 09, 10, 16 and 17 of October 2021